|
11/9/2023 0 Comments Importance of carbon cycle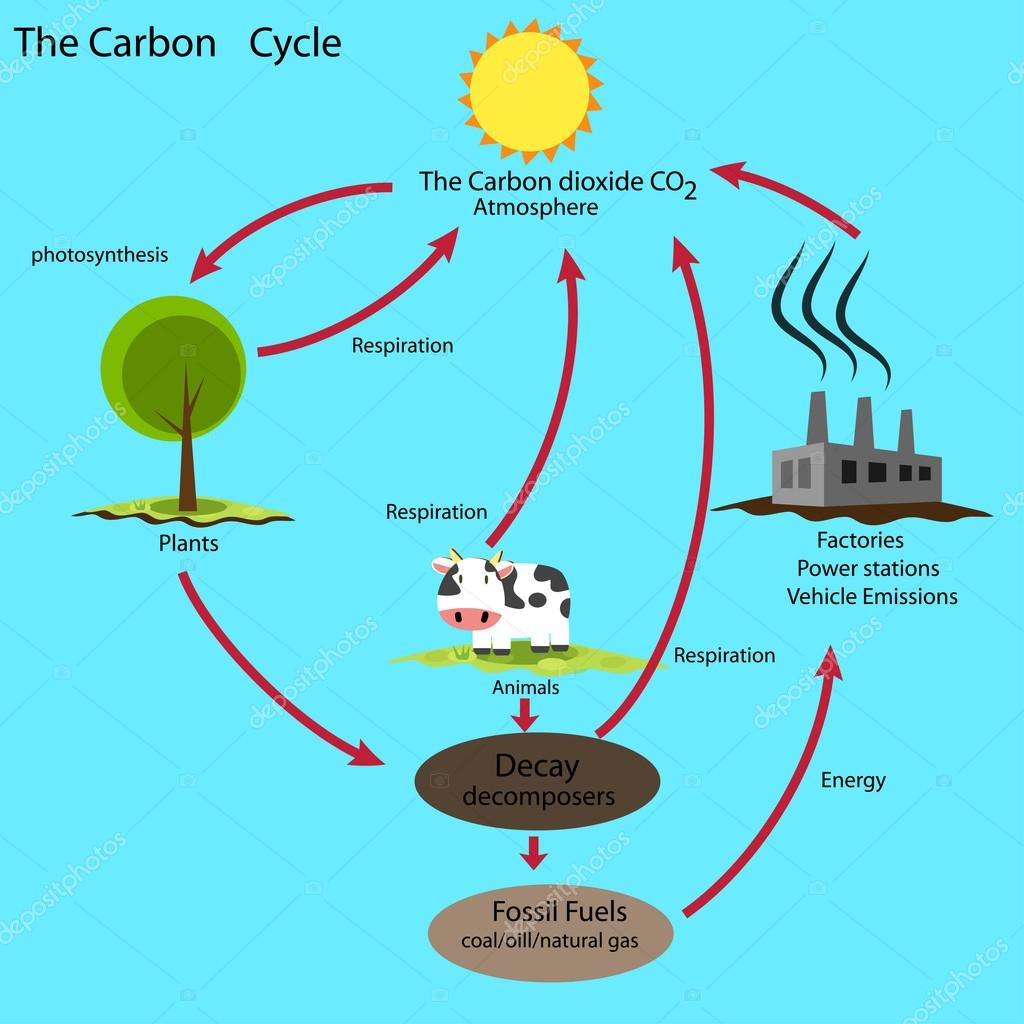
Most of the energy needed to cycle matter through earth's systems comes from the sun. Eventually, the energy is lost into space in the form of infrared radiation. As heat, it warms the planet and powers the weather system. As radiant energy, it is used by plants for food production. The one-way flow of solar energy into the earth's systems.The three main factors upon which life on the earth depends are: The earth's biogeochemical systems involve complex, dynamic processes that depend upon many factors. Changes in atmospheric circulation patterns can alter the concentration and distribution of dust ( airborne particles) that contain nitrogen (and other nutrients) for life on land and in aquatic environments.\).Regions with coastal upwelling have highly productive ecosystems because of the nutrient rich upwelled water. These nutrients can be returned to the surface by ascending currents through a process known as upwelling, which is caused by offshore winds. When organisms die they sink to the bottom of the ocean where their nutrients are released as they decay. Dissolved chemical nutrients, especially nitrogen (and phosphorus) are critical for marine organisms, including the growth of plankton and algae which form the base of most ocean food webs. Changes in ocean circulation patterns can alter the concentration and distribution of nutrients that are transported offshore.The process of producing fertilizers also introduces pollutants into the environment which alters habitats. Deforestation, habitat loss, and erosion can reduce the nutrient levels in soils.Increased precipitation can increase erosion and thus increase the transport of nitrogen (and other chemical nutrients) into soils, freshwater environments, and coastal waters.Additionally, acid rain increases chemical weathering of rocks, including manmade structures. Acid rain can affect freshwater sources, where increased nutrients can result in harmful algal blooms that reduce water oxygen levels and harm fish populations and other wildlife. The burning of fossil fuels also releases nitrogen oxides (NOx), sulfur dioxide (SO 2), and carbon dioxide (CO 2) that react with water vapor, oxygen, and other chemicals to form acid rain. The burning of fossil fuels releases nitrous oxide (N 2O), a greenhouse gas, into the atmosphere.As humans increase nitrogen soil levels, this can also reduce populations of plant species that are adapted to low-nitrogen soils. Certain species of agricultural crops, including legumes (plants in the bean family) such as soy, clover, and peas, also host symbiotic nitrogen-fixing bacteria in their roots. The abundance ( biomass) and biodiversity of bacteria, plants, fungi species that can fix nitrogen.When these algae die they are consumed by bacteria which can reduce oxygen levels in the water, killing fish and other species. For example, in aquatic environments nutrient-rich runoff ( erosion) can cause large numbers of algae to grow. 
However, increased nutrient is not always a good thing. These nutrients increase plant and algae growth. The production and use of fertilizers for agricultural activities that increase the amount of nutrients in soil or water, especially nitrogen (and phosphorus).Nitrogen levels can vary significantly in aquatic and terrestrial habitats, and can be affected by various human activities and environmental phenomena, including: Nitrogen is released back to the atmosphere by bacteria get their energy by breaking down nitrate and nitrite into nitrogen gas (also called denitrification).Ī simplified diagram showing terrestrial nitrogen cycling. Nitrogen returns to the soil when organisms release waste or die and are decomposed by bacteria and fungi. Consumers (herbivores and predators) get nitrogen compounds from the plants and animals they eat. Once nitrogen is fixed, other types of bacteria convert ammonia to nitrate (NO 3 ‑) and nitrite (NO 2 –), which can then be used by other bacteria and plants. A very small amount of nitrogen is fixed via lightning interacting with the air. This process is called nitrogen fixation. Most nitrogen enters ecosystems via certain kinds of bacteria in soil and plant roots that convert nitrogen gas into ammonia (NH 3). 
Nitrogen gas makes up 78% of the air we breathe. The largest reservoir of nitrogen is found in the atmosphere, mostly as nitrogen gas (N 2). Nitrogen cycles through both the abiotic and biotic parts of the Earth system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
